At a Glance
The OmniaSecure lead stands out as the smallest diameter defibrillation lead on the market, measuring just 4.7 French.
The OmniaSecure is the only lumenless defibrillation lead approved for placement in the left bundle branch (LBB) area.
The lead can be used for conduction system pacing, which closely mimics the heart’s natural physiology.
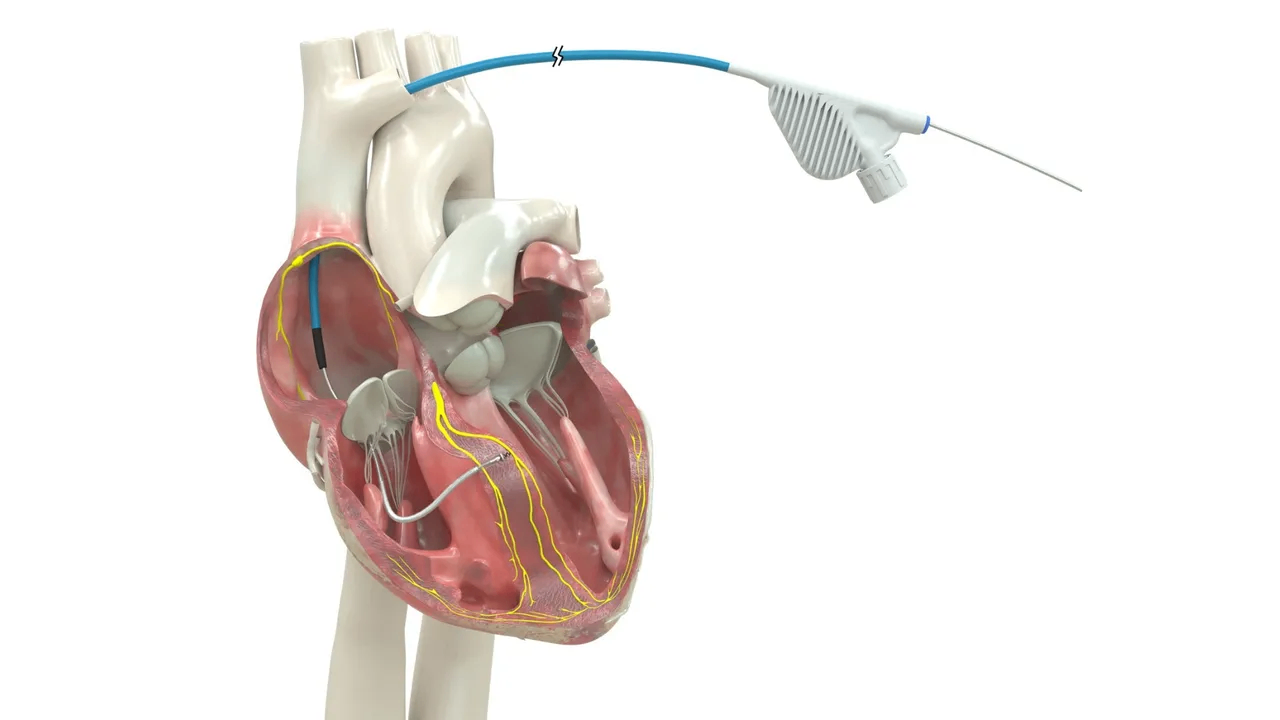
Medtronic's OmniaSecure defibrillation lead is the first lead of its kind to receive FDA approval for conduction system pacing.Image courtesy of Medtronic
Disrupting the status quo is no easy feat, especially when you're already a leader in cardiac rhythm management. Yet, Medtronic seems to have achieved just that with its OmniaSecure defibrillation lead, which recently received expanded FDA approval.
Now approved for placement in the left bundle branch (LBB) area, the OmniaSecure lead can be used for conduction system pacing (CSP), which closely mimics the heart’s natural physiology.
Additionally, patients in need of cardiac resynchronization may benefit from left bundle branch optimized cardiac resynchronization therapy (LOT-CRT), a novel therapy that combines CSP with left-ventricular pacing to further improve patient outcomes, Medtronic noted.
The OmniaSecure defibrillation lead connects to an implantable cardioverter-defibrillator (ICD) or cardiac resynchronization therapy defibrillator (CRT-D) to treat potentially life-threatening ventricular tachyarrhythmias, ventricular fibrillation (VT/VF), and bradyarrhythmias. In addition to being the first defibrillation lead approved for placement in the LBB area, Medtronic said the OmniaSecure lead is the smallest diameter defibrillation lead on the market (4.7 French, or 1.66 mm) and the only one approved for adults and adolescent pediatric patients ages 12 and up.
How Medtronic disrupted itself with the OmniaSecure defibrillation lead
Engineered based on the Medtronic SelectSecure Model 3830 pacing lead — the first pacing lead to receive FDA approval for CSP in 2022 — Medtronic said the OmniaSecure defibrillation lead is built for high reliability and durability with its lumenless construction (no hollow channel on the inside), which enables the lead to be delivered via catheter for precise placement in the right ventricle, in addition to the LBB area.
“Part of our thinking is always, you know, when you're leading, how do you then disrupt your own self? And that's sometimes a challenging thing to do because you can get caught in preconceived notions,” said Trevor Cook, VP and general manager of the defibrillation solutions business, which is part of the cardiac rhythm management operating unit at Medtronic. “But with OmniaSecure, thinking differently was important.”
Cook told MD+DI that the engineering teams often think about downsizing components of a device, but in this case, the team thought a little differently. They decided to look at the SelectSecure Model 3830 pacing lead, which Cook said is the company’s most reliable lead on the market and has been out for about 21 years, and upsizing that lead to add the components needed for defibrillation.
“So, one of the things that we needed to keep in mind as we were going through that is the defibrillation coil; that is one of the few differences on the OmniaSecure lead versus the 3830 lead. So, making sure that we had enough surface area on the defibrillation lead to defibrillate effectively at the end of the day,” Cook said. “And so, as we as we thought through that, we ended up adding a little bit of insulation to it, and it ended up turning out that the lead is a little bit bigger than the 3830 — it's at 4.7 French versus 4.1 French.”
But in doing so, he said, the company was able to demonstrate in two trials that the lead has very high defibrillation efficacy at 97.5% in the LEADR (Lead Evaluation for Defibrillation and Reliability) trial and then at 100% in the LEADR LBBAP trial (Lead Evaluation for Defibrillation and Reliability in Left Bundle Branch Area Pacing).
“I would say those are probably the two biggest notions is that notion of disrupting yourself and thinking differently and then secondly, really making sure that when you do think differently, are you taking into account the most important elements that are important to patients, like reliability and efficacy,” Cook said.
Conduction system pacing: Tapping into the heart’s natural electrical system
Conduction system pacing works by tapping into the heart's natural electrical system, providing patients needed therapy while avoiding cardiomyopathy or other complications sometimes associated with traditional pacing methods.
"Conduction system pacing is a rapidly growing therapy for patients who need a pacemaker. Now, patients who require a defibrillator and pacing have an option that can safely deliver life-saving defibrillation therapy and activate the heart's natural electrical system to enable a more synchronous, physiologic pattern," Cook said. "This approval underscores the versatility of the OmniaSecure defibrillation lead and supports its use across a variety of implant approaches to best serve a broad range of patients."
ICDs and CRT-Ds are the gold standard for preventing sudden cardiac arrest. For patients who require chronic pacing and defibrillation therapy, the OmniaSecure lead can offer treatment for dangerously fast arrhythmias while delivering conduction system pacing through the LBB area when connected to a transvenous ICD or CRT-D, such as the Medtronic Cobalt and Crome families of devices. Patients indicated for CRT may benefit when OmniaSecure is paired with a left ventricular coronary sinus lead (i.e., biventricular or LOT-CRT configurations). For patients who do not require chronic pacing, the Medtronic extravascular Aurora EV-ICD system offers protection as the only ICD with a defibrillation lead positioned outside the vascular space that also provides anti-tachycardia pacing (ATP) in a single device.
Following FDA approval for placement in traditional locations in the right ventricle, the OmniaSecure defibrillation lead launched commercially in the United States in January.





